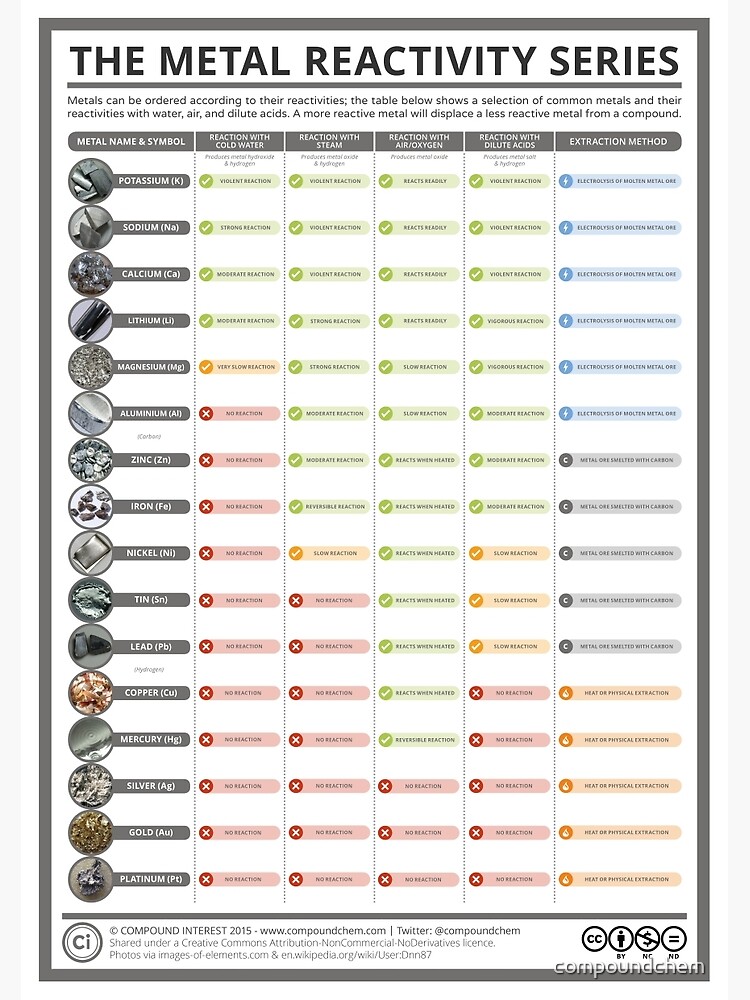
A few drops of universal indicator have been added to the water. Questionĭescribe and explain the observations when a small piece of lithium is placed on the surface of a big container of water. It is used to determine the products of single displacement reactions, whereby metal A will replace another metal B in a solution if A is higher in the series. In general, the more reactive the metal, the more rapid the reaction is. In chemistry, the reactivity seriesis a series of metals, in order of reactivity from highest to lowest. Sodium + water → sodium hydroxide + hydrogen For example, sodium reacts rapidly with cold water: The activity or electromotive series of metals is a listing of the metals in decreasing order of their reactivity with hydrogen-ion sources such as water and acids. When a metal reacts with water, a metal hydroxide and hydrogen are formed. 
Hydrogen and carbon are shown for comparison. The table summarises some reactions of metals in the reactivity series. the more easily it loses electrons in reactions to form positive ions (cations). 
In general, the more reactive a metal is: The reactivity series of metals is a chart showing metals in order of decreasing reactivity. When we complete this experiment, there are quite a few variables that we need to keep constant.When metals react with other substances, the metal atoms lose electrons to form positive ions. 
For example, magnesium metal can displace hydrogen ions from solution. We could work out the quantity of hydrogen gas produced more accurately by using a gas syringe – click here to see what the apparatus would look like if we were measuring the volume of gas produced look at method 2. An activity series is a list of substances ranked in order of relative reactivity. But if the reactivities of the metals were closer, it may be harder to see which metal was producing more hydrogen gas and therefore which metal is more reactive. The reactions of the metals with the dilute acids will also indicate how reactive the metals are and this is used to place them in the Reactivity Series. Explain how the reactivity of metals with water or dilute acids is related to the tendency of the metal to form its positive ion. A typical activity series reaction is the following: Zn (s) + Cu 2+ (aq) -> Zn 2+ (aq) + Cu (s) Which species is oxidized in this reaction Which is reduced What is the oxidizing agent What is the reducing agent The reaction actually proceeds in the direction written, rather than the reverse. This would be fine for the 3 metals that I tested because their reactivities were very different. The most reactive metals, such as sodium, will react with cold water to produce hydrogen and the metal hydroxide: 2Na + 2H 2 O > 2NaOH + H 2. 4.7.5 Atoms into ions and ions into atoms. In this experiment, we used the number of bubbles produced as a proxy for the rate of reaction and reactivity of the metals. So, the order of reactivity for these 3 metals going from most reactive to least reactive is magnesium (most reactive), iron and copper (least reactive). Copper produced no bubbles, which means that copper didn’t react at all and is the least reactive of the 3 metals. The reaction with iron produced a few bubbles, which means that iron is less reactive than magnesium, but more reactive than copper. From the diagram above, we can see that magnesium produced the greatest number of bubbles – this means that out of the 3 metals, magnesium is the most reactive.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
